Justin (left) has been paralysed from the neck down since an accident. He is now part of a clinical trial led by Prof. Dr Dr Jan Schwab (right).
Researchers in the US are testing a revolutionary drug. The hope is that it could help regrow nerves and restore bodily functions of people with spinal cord injury.
Justin is reclining in a treatment room at the Wexner Medical Center at Ohio State University. The walls are whitewashed. The room is filled with medical equipment and the unmistakable smell of disinfectants lingers in the air. Justin’s eyes are closed. His skinny legs protrude from under the bedspread. A physiotherapist standing next to him is testing whether and where Justin experiences any sensation with a safety pin.
While the physiotherapist takes notes, the young American shares his story with us: “I had an accident in 2017, in my back yard.” As so often, Justin was enjoying time on his trampoline when a spin went wrong. It didn’t really look serious, but the young college student fell unfortunately and fractured his cervical spine. An injury to his spinal cord left him paralysed. He can’t walk, can barely feel his body, and has restricted hand movement capability. What troubles him most, however, is that the spinal cord injury determines every aspect and second of his life. “I miss being able to be ‘last minute’,” he says. “I have to plan so many things throughout my days. Before, if I needed to be somewhere, I could just wake up, roll out of bed, get a bite to eat and get there really easily.”
Interceptor molecule promotes nerve growth
Justin has come to the Wexner Medical Centre to participate in a clinical trial. The trial involves testing a drug that is designed to help people such as himself: patients who suffer from chronical spinal cord injury, i.e. have been paralysed for more than a year. The drug is supposed to ensure regrowth of affected nerve fibres, thus restoring important bodily functions to the patients. Yet is that even possible?
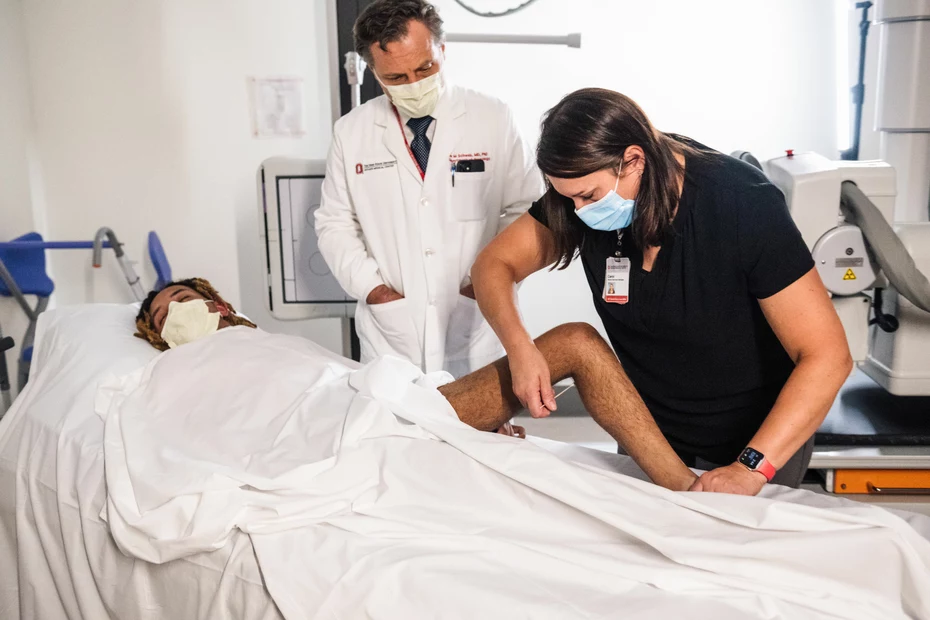
Safety pins, cotton swabs, and touch pens are used to determine whether the patient can feel anything.
“It is,” says study initiator Stephen Strittmatter. The Yale professor has spent the last few decades working hard on solving the puzzle of how to encourage damaged nerves to resprout. “When someone suffers a nerve damage, it interrupts the fibres between the nerve cells but leaves a lot of them alive. We need to reconnect them, to re-establish the neural network”, he explains. This cannot be achieved by natural means. The damaged nerves actually seek to regrow, but several proteins prevent them from doing so. “We discovered a receptor for three key proteins and developed an interceptor molecule called the ‘Nogo trap’,” explains Strittmatter. This Nogo trap shields the receptor of the nerve fibre from being detected by the proteins. The result: the nerve fibre can regrow and form new connections.
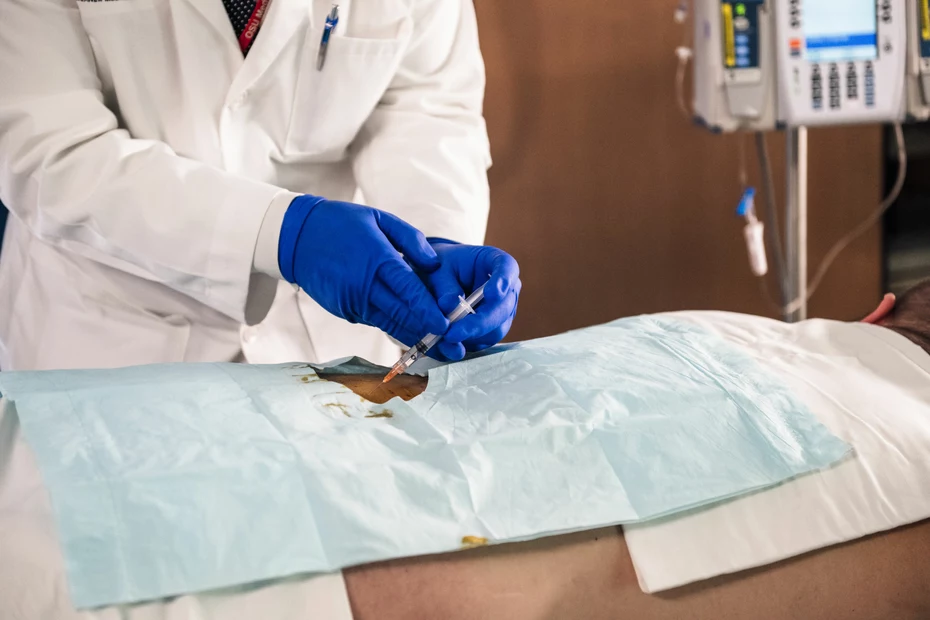
The “Nogo trap”is injected into the spinal fluid.
Injection into the spinal fluid
This approach has already yielded great success in experiments. When Strittmatter injected the Nogo trap into the spinal fluid of paraplegic rats, a third of the paralysed animals regained full movement. Following these successful preclinical results, the Nogo trap has been undergoing a clinical trial involving people with spinal cord injury led by Strittmatter since 2019. The aim is to determine whether the drug is also safe and effective in humans. The Wexner Medical Center is one of six institutions partaking in the study. Professor Jan Schwab is in charge of conducting the study there. He has monitored Justin and other participants over the last few months and explains the procedure: “The patients receive several infusions. We inject the medication directly into the spinal fluid through the lumbar spine. This allows it to directly target the injury site and impact the entire spinal cord.”

Motor skills tests determine whether a patient can turn a key in a lock.
Then, numerous tests determine whether the patients are making progress in their ability to move and feel. That’s why Justin spends from nine in the morning to four in the afternoon at the clinic. Safety pins and cotton swabs are used to evaluate whether his sensory capacity has improved. This is followed by tests involving arm strength, everyday situations, and fine motor skills. Has his grip developed? Can he insert and turn a key in a lock? Can he pour himself a glass of water independently? The findings are meticulously recorded. They will later enable researchers to draw conclusions as to whether new nerve connections have really formed.

Prof. Stephen Strittmatter (left) is the initiator of the clinical trial, Prof. Dr Dr Jan Schwab (right) is leading the trial in Ohio.
What is spinal fluid?
The spinal fluid (or cerebrospinal fluid) surrounds the spinal cord and protects it from shocks.
High hopes
"It's a complicated study that is highly demanding on everyone involved", Strittmatter explains. “If successful, however, it would open the door to an entirely new class of drugs.” Schwab has high hopes, too: “This study is really unique. If it succeeds as hoped, it will represent a major milestone.”
The two neurologists are still uncertain whether their research will be rewarded with success. First results indicate that the drug is safe and well-tolerated. It is also important to bear in mind that the study is blinded. This means that some patients receive an infusion with the drug (Nogo trap), while others merely receive a placebo. This is the only way researchers can gather scientifically relevant data. Even Justin doesn’t know what kind of medication they injected into his back. Yet that’s not his main motivation. He understands that the findings generated for spinal cord research are invaluable. “I’m taking part in the study because I want to help,” he explains. “Besides, it fills me with hope.” The hope that research will progress and one day result in a cure. What would he do first when the time has come? “If I could ever walk again, I would probably go dancing somewhere. Dance with my girlfriend. That’s probably the first thing I would do.”
How Wings for Life Foundation supports
Wings for Life started funding Strittmatter’s research project in its early stages and has subsequently successfully accompanied it into the clinical testing phase. The foundation has provided the study with seven million US dollars in funding – its highest funding amount to date.
What is the current status?
In this study, the safety and tolerability of the applied drug were tested. Initial encouraging data show that no serious side effects occurred. Currently, all data from this study are being evaluated and compared with each other. The final results are still pending.

Participation in the study gives Justin hope.
Image credits: Michale Millay
